Sodium element valence electrons9/12/2023 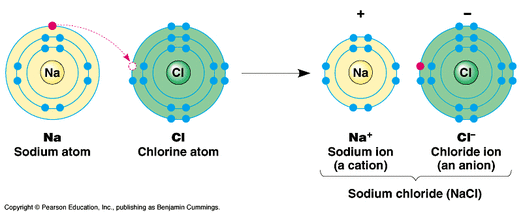
Starting with lithium (Li), which has one valence electron, we can move across horizontally and add one valence electron each time as follows,įurthermore, every atom on the periodic table below lithium (Li) will also have one valence electron (i.e. If we start by looking at the second row, and follow along horizontally, we can assign valence electron numbers to each element. The easiest way is to use the periodic table as your guide. There are a number of ways to approach this question. (Quick note: In the periodic table, rows are horizontal lines, rows are vertical lines.) For fluorine, which is in the second row, the valence electrons will be found in the second shell. For sodium, which is in the 3rd row of the periodic table, the valence electrons will be found in the 3rd shell. The shell number representing the valence shell will differ depending on the atom in question. Valence electrons are electrons found in the outermost shell of an atom. Here is a video which gives further explanation of this topic. Fluorine has the electron arrangement 2.7. The last number is how we know the number of valence electrons.Īluminium has the electron arrangement 2.8.3. 
So, sodium's 11 electrons are arranged this way: 2 electrons in the first "shell", 8 electrons in the second "shell" and 1 electron (the valence electron) in the third "shell". The third "shell" is a bit more complicated but let's just say that it takes up to 8 electrons as well (for now.). The second "shell" can have up to 8 electrons. Depending on your level of Chemistry, it is probably easier to think of them as particles orbiting the nucleus. Sodium has 11 electrons: its atomic number is 11, so it has 11 protons atoms are neutral, so this means sodium also has 11 electrons.Įlectrons are arranged in "shells" or energy levels. As a source of sodium in the laboratory.Valence electrons are the outermost electrons, and are the ones involved in bonding. Sodium hydroxide is also used to make caustic soda and bleach, as well as other industrial products.ġ0. In the production of sodium hydroxide, which is used to make soap in the process of purifying brine and in water treatment. As a strong reducing agent in the chemical industry.ĩ. In the production of sodium chloride, which is used in the production of table salt, water softeners, and other industrial products.Ĩ. In the production of sodium bicarbonate, which is used in the food and beverage industry.ħ. As a de-icing agent on roads and pavements.Ħ. In the production of sodium carbonate, which is used in the production of paper, textiles, and other industrial products.ĥ. As a flux in the metal smelting industry.Ĥ. Sodium helps to produce hard water soap and is also used in the manufacture of detergents.Ģ. In the production of soaps and detergents. Some of the most common uses of sodium metal include:ġ. Sodium is a very versatile element and has many uses in both industry and everyday life. The ocean waters are chock full with this mineral – many scientists believe they’re even spilling out more than what goes in due to climate change happening underwater. You’ll never see it in its pure form because of how reactive and volatile it is, but we do have compounds like table salt or NaCl (salt-chlorine) to thank for our ability to get at all that goodness inside us! It’s the sixth most abundant element on Earth. That’s because only one out of twenty known varieties will maintain its stability, while the others decay over time and become more likely to change back into their original element. Soda ash also known scientifically as “common lime” may sound like some kind of weird science experiment gone wrong however, its discovery helped revolutionize modern agricultural practices because before then farmers were using animal bones or other sources containing calcium carbonate which limited crop yields due to too rapid spoilage during storage. It has the ability to float on top of the liquid, but when mixed together they produce an explosion of gas as well! Sodium’s most famous compounds include table salt (NaCl), sodium nitrate(sodium-potassium), and baking soda – all three are found abundantly around us every day without even realizing how many uses there really are for them. Sodium is a reactive metal that can easily be cut with a knife and will burn if you put it in water. In 1807, he conducted an experiment to isolate this now important chemical by applying electrolysis on caustic soda which resulted in its isolation and identification as a new element – with Davy himself going down into history books for making such an impactful contribution! Characteristic properties of Sodium metal Characteristic properties of Sodium metalĮnglish chemist Sir Humphry Davy is often remembered as the man who discovered sodium.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
